Electrolysis
|
Electrolysis is used to create chemical reactions that usually wouldn't happen with a direct current of electricity. This technique is done in an electrolytic cell which contains a solution or molten substance which is called an electrolyte. At the two ends there is an anode which is the positive part and a cathode which is the negative part. The two conductors (the anode and cathode) will have a direct current of electricity sent through them and that therefore starts the process called electrolysis. This is used to refine metals, getting metals out of other metal ores, electroplating and anodizing.
|
Diamagnetism
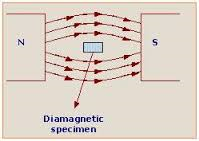
Diamagnetism is when two of the same charge repel each other. Like when the same colour or sides of a magnet repel instead of attract. This is because the two things are the same charge, for example 2 negatively charged things will repel each other. Diamagnetic atoms always have their electrons all in pairs. When two electrons share the same orbital, their spin numbers must be different. When two electrons are in a pair, their spin number is 0 and they become diamagnetic electrons. Atoms with diamagnetic electrons only are called diamagnetic atoms.
Paramagnetism
Paramagnetism is the opposite of diamagnetism. It is when two oppositely charged conductors attract to try and neutralise the charge. It is why the red end of a magnet sticks to the blue end but not the other way around. Paramagnetic atoms have at least one unpaired electron. When an electron is not in a pair it is called a paramagnetic electron. No matter how many diamagnetic electrons there are, as long as there's one paramagnetic electron the atom will be a paramagnetic atom.